Mass cytometry, or CyTOF, is a powerful technology that has been used for comprehensive multiparameter characterization of immune cells. We recently conducted a 38-parameter characterization of HIV entry and productive infection of tissue CD4+ T cells by CyTOF, and used an analytical approach that takes advantage of the high-dimensional nature of CyTOF datasets to distinguish receptors modulated during infection from those differentially expressed on preferentially infected cells. These studies identified a subset of memory CD4+ T cells susceptible to HIV entry but not productive infection. Ongoing work in the lab seeks to characterize the molecular basis of this post-entry restriction, to use CyTOF and a variety of high-dimensional CyTOF data analysis tools to characterize the types of cells that are productively and latently infected with HIV, and to apply the high-dimensional analysis approaches we have developed on HIV-infected cells characterized by single-cell RNAseq. We are also working closely with UCSF’s SCOPE cohort to identify signatures and biomarkers of infected cells that persist in HIV-infected individuals on suppressive antiretroviral therapy, as well as signatures of immune effector cells that can control viral replication in these infected individuals.
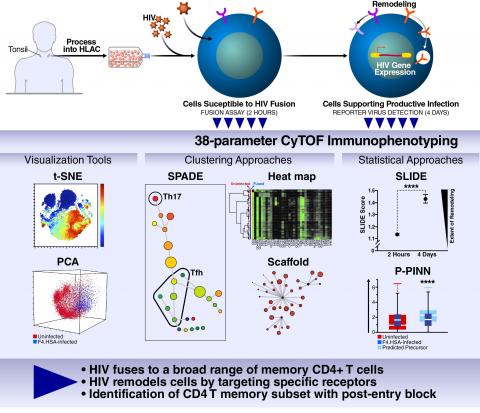
Schematic illustrating the steps involved in characterizing the susceptibility of tissue-derived CD4+ T cells to HIV infection. Tonsils were processed into human lymphocyte aggregate cultures (HLACs), infected with an HIV-1 reporter virus, and then monitored for HIV entry after 2 hours or productive infection after 4 days. Using a variety of visualization, clustering, and statistical approaches, the susceptibility of different subsets of CD4+ T cells to HIV entry and productive infection was assessed, which led to the discovery of a new subset of memory CD4+ T cells highly susceptible to HIV entry but not productive infection. We are currently using these analytical approaches to characterize how HIV changes the properties of CD4+ T cells upon infection, including studies using patient cells.
